ABSTRACT
The article presents the results of a survey of the water mite fauna in 45 dam reservoirs in the Czech Republic that are sources of drinking water. In 37 reservoirs, the average seasonal concentration of chlorophyll a in water were lower than 20 µg/L, which indicates that most of these reservoirs have an oligotrophic to slightly mesotrophic character. Samples were taken with a hand plankton net in the stony littoral at a depth of 0.5–1.0 m at all localities; therefore, it is possible to compare findings from individual localities, although they do not represent quantitative data of water mites related to unit area or water volume. In total 1,356 water mites (849 adults, 507 nymphs) were caught and 34 species were recorded. Twelve species occurred in more than 15 % of the investigated reservoirs and accounted for 87.4 % of all individuals caught. These species show great locomotive activity, they are adapted for swimming/floating, and they commonly occur in the littoral fauna of European lakes. The most numerous species were Brachypoda versicolor (Mueller, 1776) and Unionicola crassipes (Mueller, 1776), which occurred at the largest number of localities (23 and 21) and totalled 357 individuals, which is 26.3 % of all recorded water mites. Fourteen species of water mites occurred only in one locality; these are common in standing waters, and their preferred habitat is littoral vegetation. On the other hand, species of the genus Lebertia Neumann, 1880, were not found in the monitored reservoirs: they form a stable component of water mite fauna in lakes, especially at greater depths. Forelia longipalpis Maglio, 1924 is a new species for the fauna of the Czech Republic; Arrenurus albator (Mueller, 1776) and Hygrobates trigonicus Koenike, 1895 have previously been recorded in the Czech Republic, but have not yet been listed in the species database of the Nature Conservation Agency of the Czech Republic (NCA). The results provide the first more extensive knowledge of the water mite fauna in reservoirs in the Czech Republic, as until now most hydrachnological studies in the Czech Republic have been focused on the water mites of ponds and watercourses.
The study was created with the support of the NCA for the implementation of the Species Occurrence Database under a contract for the development of the work Diversity of water mites in water reservoirs of the Czech Republic in 2025.
INTRODUCTION
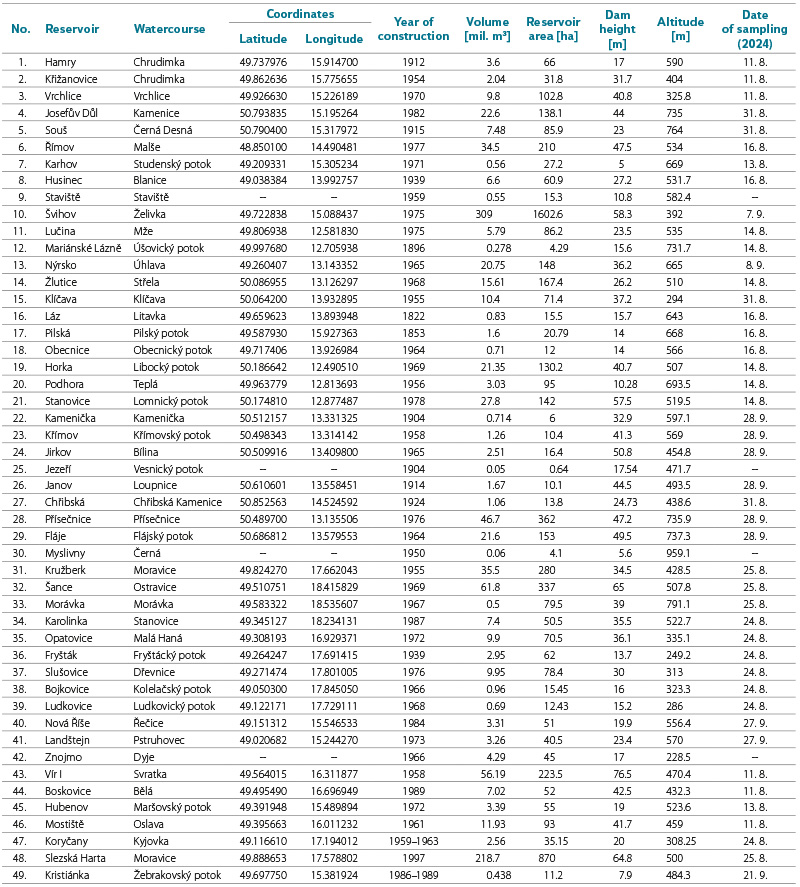
Previous research on water mite fauna in the Czech Republic has focused mainly on ponds, running waters (especially streams), and springs; the occurrence of water mites in dam reservoirs has received little attention, although there are 165 significant dam reservoirs in the country. Of these, 47 are designated by Decree No. 137/1999 Coll. [1] for water-supply purposes and provide 50 % of the drinking water supply for the population of the Czech Republic. The management of these water reservoirs and land-use practices within their catchments are subject to protective regimes, not only to ensure a sufficient volume of stored water to meet supply demands, but above all to maintain the highest possible water quality. Protection zones of water-supply sources (see Act No. 254/2001 Coll., on waters and on amendments to certain laws, as amended [2]) and regulated agricultural management within catchments (in accordance with river basin management plans under the Water Framework Directive 60/2000/EC) strengthen the provision of high-quality raw water for water treatment. A total of 36 water reservoirs (Tab. 1) are located at elevations above 400 m a.s.l., and their inflows consist of upper (headwater) sections of watercourses, i.e. catchments without significant sources of pollution. For these reasons, water quality in water reservoirs is significantly higher than in multi-purpose reservoirs, ponds, and smaller standing waters. At the same time, biomanipulation is applied in these reservoirs through the support of so-called purpose-oriented fish stocking, with the aim of limiting phytoplankton development by influencing the structure of the food web within the reservoir biocoenosis. Increased stocking of predatory fish species affects the food chain by reducing the abundance of small fish species, which allows the development of larger zooplankton species (especially cladocera). Through their predatory activity on planktonic algae, these organisms can regulate phytoplankton biomass even under conditions of elevated nutrient concentrations.
Tab. 1. List and characteristics of water reservoirs (in order according to Decree No. 137/1999 Coll. [1]) and date of water mites sampling in 2024
To date, only a few isolated records exist on the occurrence of water mites in dam reservoirs in the Czech Republic, mostly obtained as part of hydrobiological research projects (e.g. [3]). Similarly, in other European countries, little attention has been paid to the water mite fauna of reservoirs; most information on standing waters comes from studies of lakes and ponds.
The aim of this survey was to obtain data on the water mite fauna in dam reservoirs in the Czech Republic, with a focus on their occurrence in the littoral zone over the stony surface of the dam embankment, i.e. in that part of the reservoir which is connected to the pelagic zone, is not overgrown with littoral vegetation, and has characteristics of certain lacustrine habitats, thus allowing comparison with findings of water mites from European lakes.
SAMPLING SITES
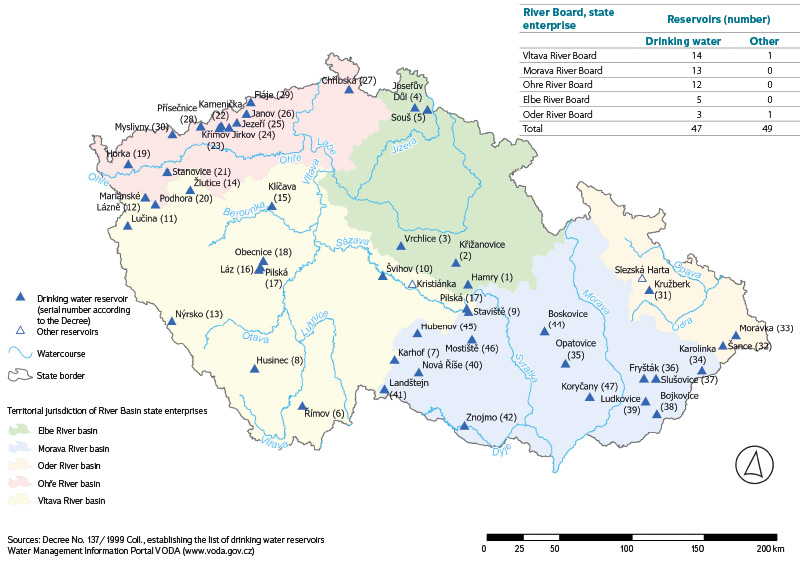
The distribution of water reservoirs in the Czech Republic is shown in the map in Fig. 1. A list of them is provided in Tab. 2, which contains basic information on the individual reservoirs from which water mite samples were collected in August and September 2024. The majority of water reservoirs (80 %) are located at elevations above 400 m a.s.l.
Fig. 1. Location of investigated reservoirs in the Czech Republic, indicating the responsibility of the river basin administrators (River Boards, state enterprises) that manage the reservoirs (Source: Ministry of Agriculture, elaborated by Mgr. Monika Stádníková)
Tab. 2. List and additional characteristics of investigated water reservoirs (water transparency measured by Secchi disc, chlorophyll a concentration, water level fluctuations; data provided by colleagues of River Boards, state enterprises) and numbers of water mites found
Of the total of 47 water reservoirs listed in Decree No. 137/1999 Coll. [1], sampling was not carried out at Znojmo reservoir (which is essentially a multipurpose reservoir with water-supply abstraction) or at Staviště reservoir (access to the dam was closed and it was not possible to obtain a sample from a suitable location). Jezeří reservoir is also missing (it was drained in the given year), as is Myslivna reservoir, which, given its size, has more the character of a pond. On the other hand, sampling was carried out at Slezská Harta reservoir, which serves as a “pre-reservoir” for Kružberk reservoir, from which water is abstracted for supply purposes. A sample was also taken from Kristiánka water reservoir near Světlá nad Sázavou, which is a permanent source for a public water supply operated by Vodovody a kanalizace Havlíčkův Brod, a. s. In total, water mite fauna samples were collected from 45 reservoirs; the numerical designation of the sites/reservoirs follows their order in the cited Decree, with the two additional reservoirs assigned the numbers 48 and 49 in the list.
Sampling sites were located in the littoral zone immediately adjacent to the dam structures or over the stony bottom at the toe of the dams (geographical coordinates are given in Tab. 1); examples of several specific sites are shown in Figs. 2–9.

Fig. 2. Hubenov reservoir (site no. 45), view from the dam to the sampling site
Fig. 3. Přísečnice reservoir (site no. 28), view of the dam and sampling site
Fig. 4. Horka reservoir (site no. 19), view of the dam and sampling site

Fig. 5. Kružberk reservoir (site no. 31), view of the dam and sampling site

Fig. 6. Karolinka reservoir (site no. 34), view from the dam to the sampling site

Fig. 7. Mostiště reservoir (site no. 46), view of the dam and sampling site

Fig. 8. Vír reservoir (site no. 43), view of the dam and sampling site

Fig. 9. Nová Říše reservoir (site no. 40), view of the sampling site
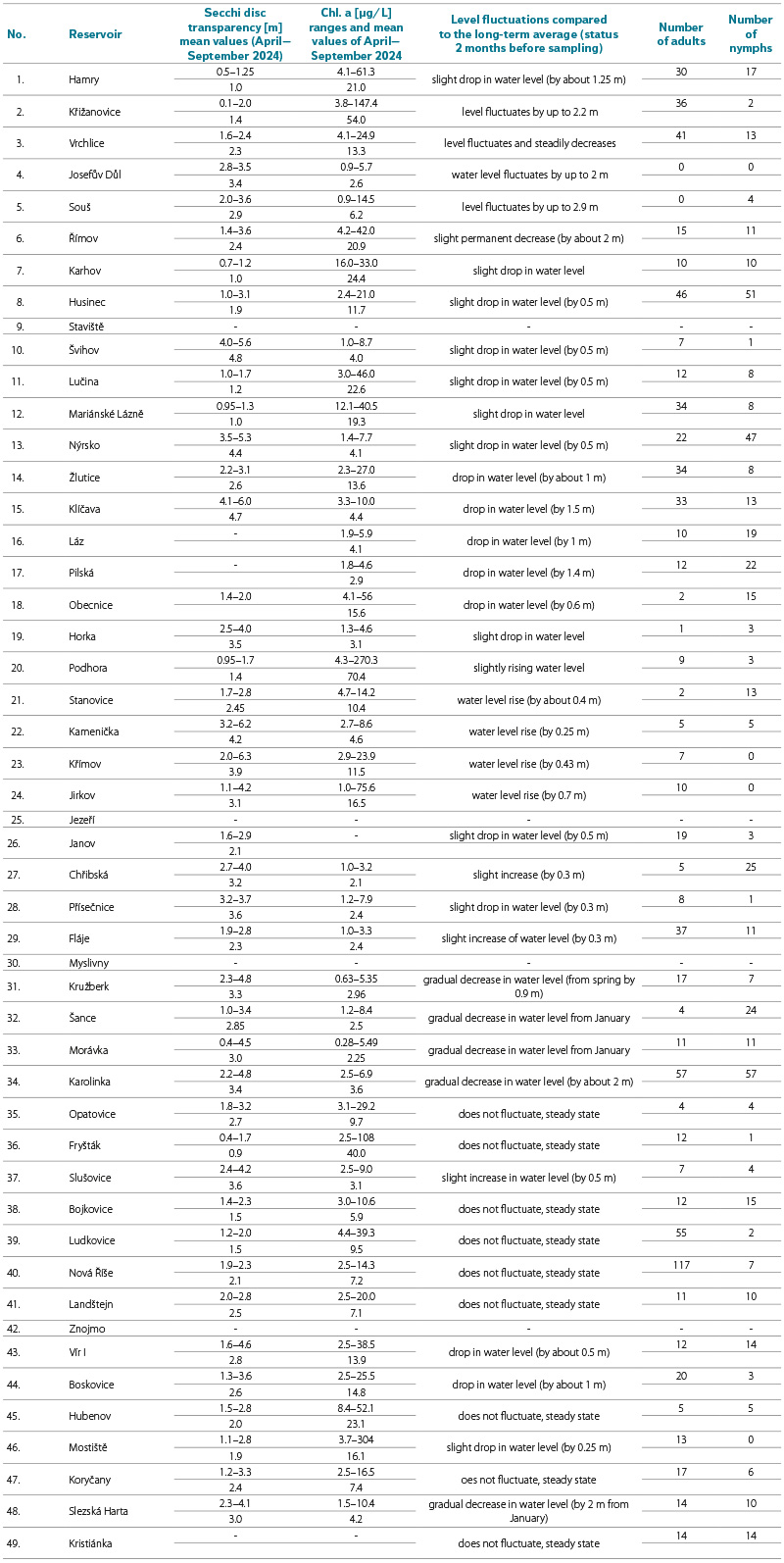
Tab. 2 contains data on chlorophyll a concentrations in the individual reservoirs, which allow comparison of their trophy, as the quantity of phytoplankton is an indicator of nutrient loading in water bodies. Mean values and ranges of chlorophyll a concentrations are presented, based on samples collected at weekly intervals from April to September 2024. Typically, 9 to 11 values were determined over the course of the season. Tab. 2 includes data on water transparency, also measured at weekly intervals during the period from April to September. These data were provided by staff of the River Boards, state enterprises from ongoing long-term monitoring of water quality in all water reservoirs.
From water-level fluctuation data obtained from monitoring of the individual reservoirs (provided by the respective River Board, state enterprise), it was confirmed for each site that the sampling locations had been submerged for more than three months prior to sampling; thus, the water mite fauna samples were collected from long-term submerged surfaces of the littoral zone. In cases of a gradual long-term decrease in water level by several metres, for example in Horka reservoir (site No. 19), changes in benthic community composition in the littoral zone cannot be ruled out.
METHODOLOGY
For sampling water mite fauna, a standard method was used involving the collection of organisms with a plankton net mounted on a pole; the diameter of the circular opening was 30 cm and the mesh size was 0.350 mm. Each sample was taken by sweeping the net along the dam face or over the stony riprap at the toe of the dam, with horizontal movement of approximately 1.5 m and at depths of 0.5–1.0 m below the water surface for a duration of two minutes. Sampling sites were selected so that the substrate (stony surfaces under the open water column) was largely free of vegetation and morphologically very similar (Figs. 2–9).
This methodological approach does not provide an absolute quantification of the number of individuals collected, i.e. in relation to water volume or surface area; however, it allows comparison of the number of water mites captured, the frequency of species present, and their occurrence within the same habitat (littoral over a stony substrate). It therefore represents a semi-quantitative method for assessing the occurrence of water mites at individual sites, making it possible to express the relative abundance of the recorded species.
The collected material, consisting of detritus with zooplankton, meiobenthos (including water mites), macrozoobenthos, and larger phytoplankton species, was transferred from the net into water in a plastic tray (30 × 50 cm). Water mites were picked out on site using a pipette and fixed in Koenike’s solution (a mixture of glacial acetic acid, glycerine, and distilled water [4]). After the completion of sampling at the end of September 2024, the individual water mite samples were sorted under a stereomicroscope (at 10–50× magnification), and both adult specimens and nymphs were counted. From the adult specimens, already sorted to genus level, several individuals from each morphologically uniform group were selected for further preparation. After heating in potassium hydroxide (30 %), during which the chitinous body structures turn brown, the homogeneous body contents are expelled from the individual specimens under a stereomicroscope; after rinsing in water, the specimens are transferred to glycerine. Subsequently, on a microscope slide, the organs necessary for species identification are prepared (mouthparts are separated, and in some cases certain legs). After embedding in glycerine jelly and covering with a coverslip, permanent slides of adult specimens were prepared, suitable for microscopic identification to species level. These permanent preparations serve as reference material and allow the acquisition of photographic documentation. Glycerine jelly is a historically well-established medium for zoological microscopic preparations and is still used today for preserving voucher material of water mites, see [5].
Immature stages of water mites (nymphs) were, with few exceptions, not prepared, as their morphology generally does not allow reliable identification to species level.
Species identification of water mites was based primarily on literature published in the last 10–12 years [6–8]; however, older identification keys are also useful for certain more detailed information [4, 9].
RESULTS
Average chlorophyll a concentrations (Tab. 2) during the growing season were below 10 µg/L in 26 reservoirs, while in 11 reservoirs they ranged between 10 and 20 µg/L. Only six reservoirs exceeded 20 µg/L, in three cases very substantially. These data indicate that the sites are predominantly oligotrophic to slightly mesotrophic, which corresponds to the requirements for water quality of sources used for drinking water treatment. This is also reflected in the average values of water transparency during the summer (growing) period, which ranged between 2 and 3 m; only in a few cases were they less than 1 m.
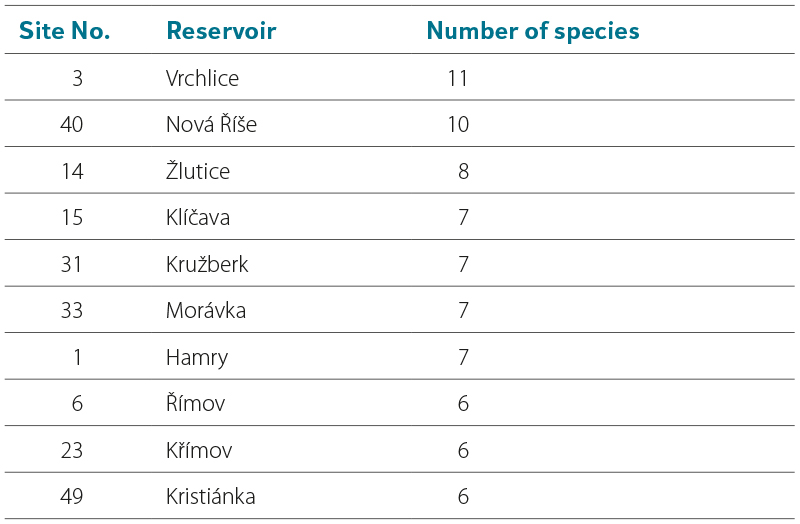
A total of 1,356 water mites were recorded in 45 water reservoirs, of which 507 specimens (37.3 %) were immature stages (nymphs), for which identification to species level is uncertain (Tab. 3). The list of water mite species in Tab. 3 is therefore based solely on the identification of 849 adult individuals, whose occurrence and abundance were found at the individual sites.
Tab. 3. Number of adults and nymphs of individual water mite species found in investigated reservoirs
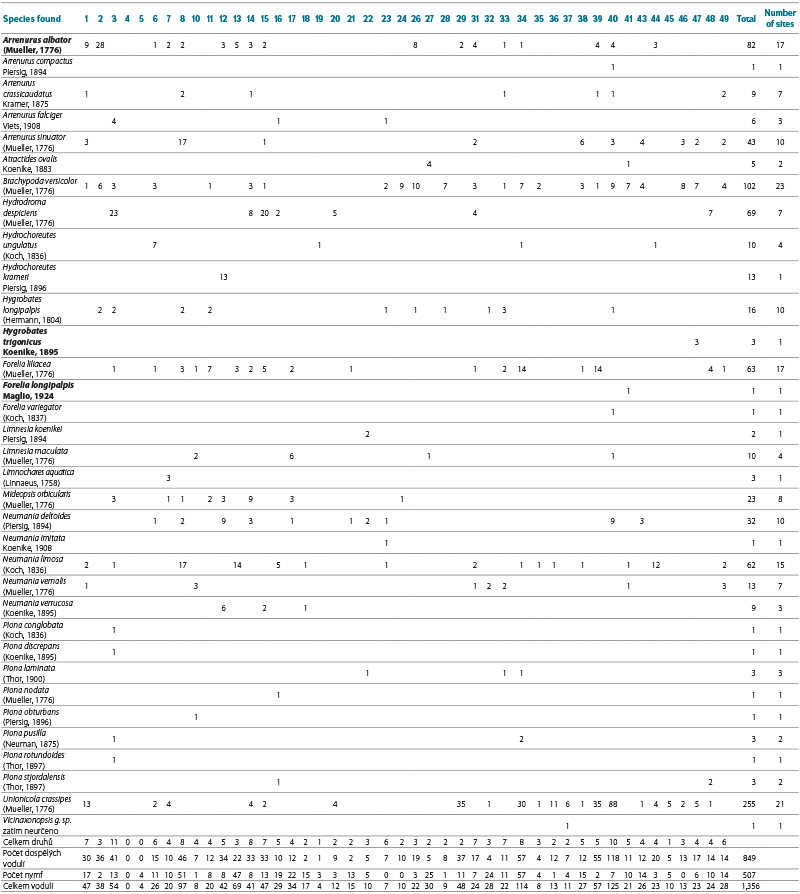
The numbers of water mites vary considerably among individual sites, as does the proportion of immature stages (nymphs). Tab. 4 presents data from sites with more than 40 water mites (adults and nymphs), i.e. more than 3 % of the total number of collected water mites. The highest numbers of water mites were found in Nová Říše reservoir (site No. 40), Karolinka reservoir (site No. 34), and Husinec reservoir (site No. 8). By contrast, no water mites were found in Josefův Důl reservoir (site No. 4), and only four nymphs were recorded in Souš reservoir (site No. 5) (only one was identified, belonging to the genus Hydrochoreutes Koch, 1837). The likely cause is the low pH values of the water in these reservoirs (see Discussion). Only a single water mite was also found in Horka reservoir (site No. 19); a probable reason may be the long-term gradual but substantial decrease in water level of approximately 6–8 m (as shown in Fig. 4), which may have significantly reduced the availability of organisms necessary for the development of water mite larvae. A total of 34 water mite species were recorded across all 45 studied reservoirs. Twelve species dominated in both occurrence and number of individuals, accounting for 87.4 % of all recorded adult water mites (Tab. 5). All these species possess numerous swimming setae on their legs, and Unionicola crassipes (Mueller, 1776) as well as species of the genus Neumania Lebert, 1879 are characterised by very long legs, as illustrated in Microphotographs I–L. The number of species recorded in individual reservoirs is given in Tab. 3; the highest numbers were found in Vrchlice reservoir (site No. 3) and Nová Říše reservoir (site No. 40), see Tab 6. Fourteen species occurred at only one site, while two to three species were recorded at three sites. These are species of the genera Arrenurus Dugès, 1834, Limnesia Koch, 1836, and Piona Koch, 1842, which otherwise commonly occur in large numbers in standing waters with macrophyte vegetation, especially in ponds.
Tab. 4. Number of adults and nymphs of individual water mite species found in investigated reservoirs
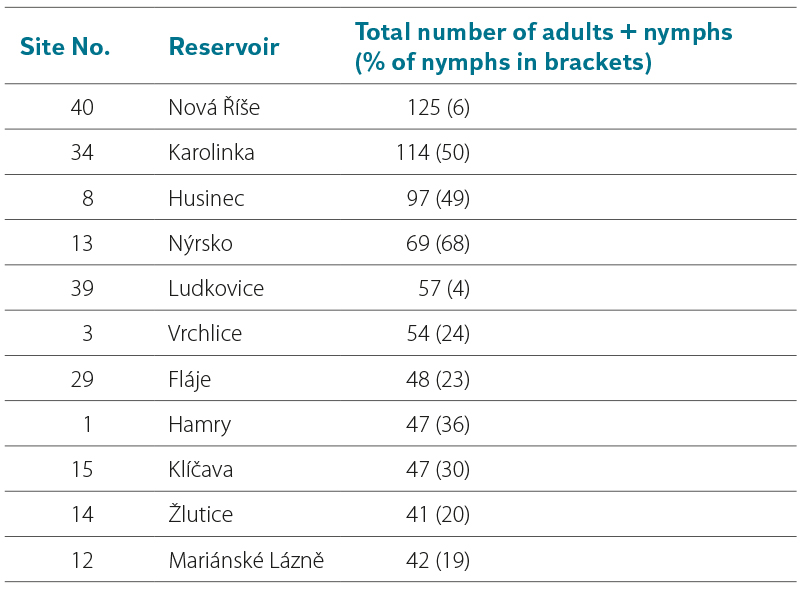
Tab. 5. Species of water mites with the highest abundance in all investigated reservoirs
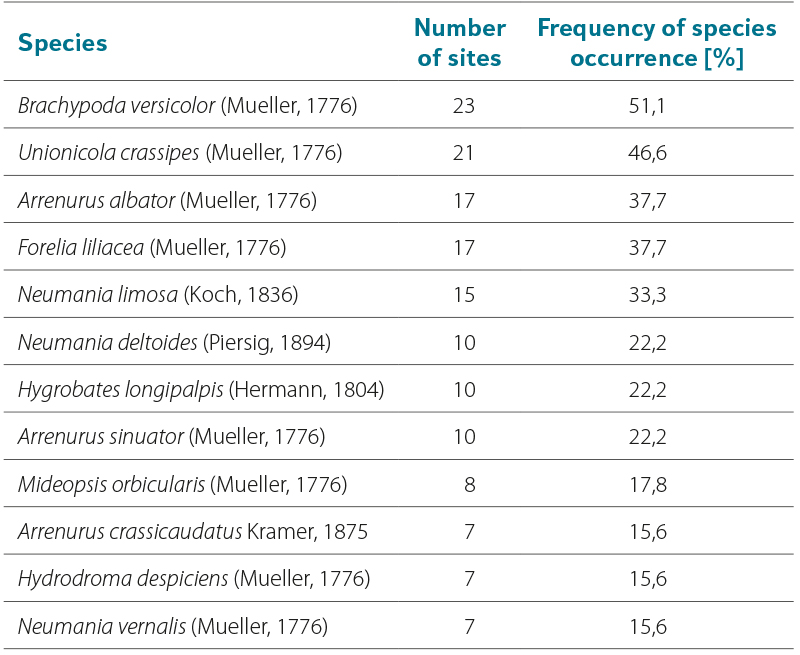
Tab. 6. Reservoirs with the highest number of water mite species
Three recorded species expand the list of water mite species documented in the Czech Republic in the database of the Nature Conservation Agency of the Czech Republic. These are the following species:
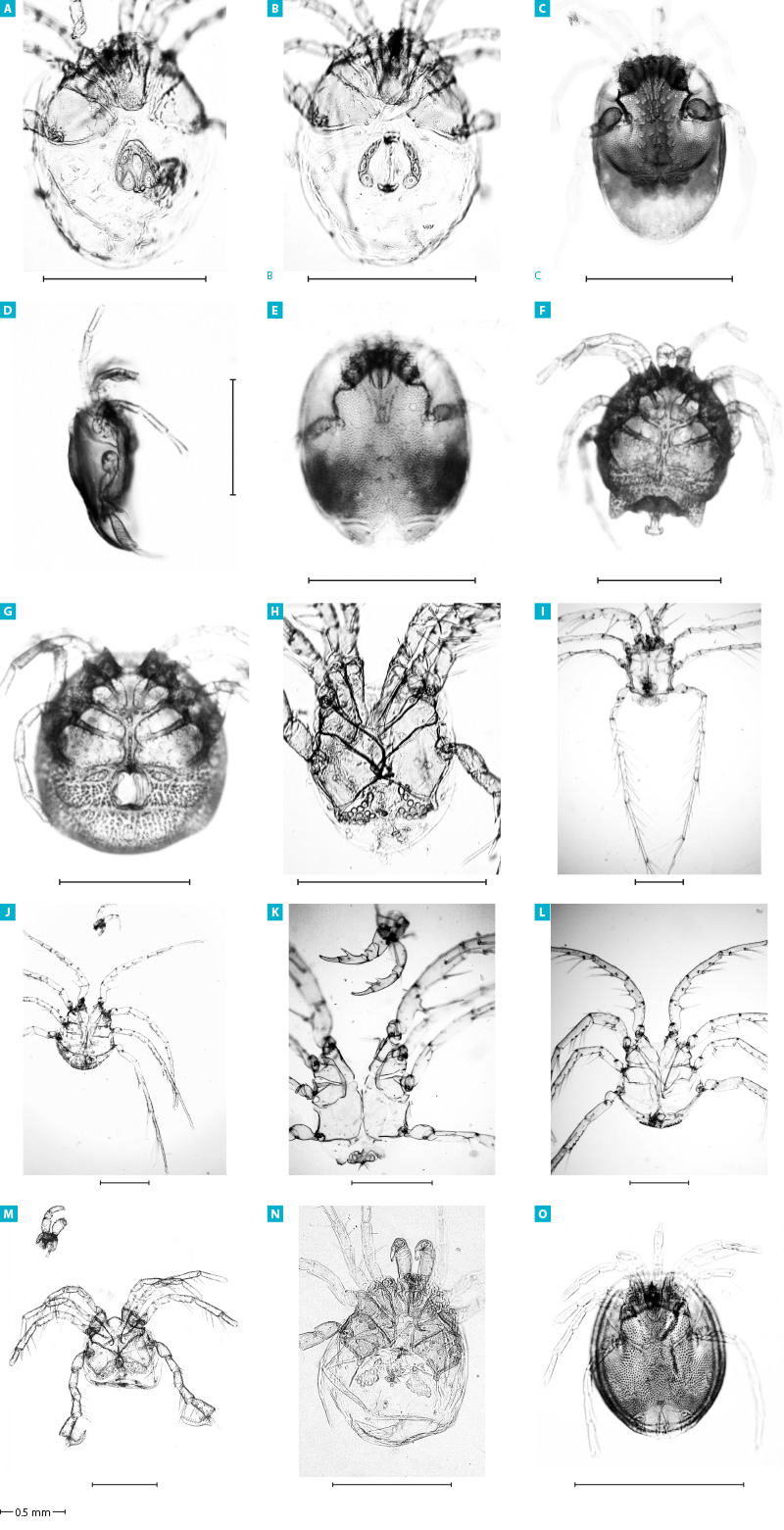
- Forelia longipalpis Maglio, 1924 (Microphotograph H)
The record of this species in Landštejn reservoir (site No. 41, see Tab. 3) also represents a species new to the fauna of the Czech Republic, as it has not previously been reported in published studies on water mites in Czech waters. - Arrenurus albator (Mueller, 1776) (Microphotographs F, G))
This species has been recorded previously, and Láska [9] reports it as common throughout the Czech Republic. It is among the most frequently recorded species of the genus Arrenurus Dugès, 1834 in European water bodies, especially in large lakes [10, 11]. - Hygrobates trigonicus Koenike, 1895 (Microphotographs A, B)
This species is not new to the fauna of the Czech Republic either; however, it is reported as a “relatively rare species” from submontane streams [9]. It was found in the studied reservoirs but is common in lakes [11, 12]. - The record of Atractides ovalis Koenike, 1883 is also noteworthy, as it is the only species of this genus that occurs in standing waters and is particularly common in lakes of northern Europe [10].
The identification of the water mite designated as “Vicinaxonopsis Cook, 1974” remains unresolved. It belongs to the subfamily Axonopsinae, Viets, 1929. The genus Vicinaxonopsis has so far been described only from Bulgaria and Sardinia [7]. Only a single specimen (Microphotograph O) was found in Slušovice reservoir (site No. 37, see Tab. 3), and further clarification of this record will therefore require repeated sampling at this site.
The article also includes microphotographs of water mite species that expand the existing list of species recorded in the Czech Republic by the Nature Conservation Agency, as well as species most frequently occurring in water reservoirs and species found only sporadically in the country (Microphotographs A–O).
DISCUSSION
Water mite fauna in dam reservoirs in the Czech Republic has not been studied in detail to date, and available knowledge is therefore only fragmentary.
The first data on the occurrence of water mites in a dam reservoir in the Czech Republic (then Czechoslovakia) were published in 1960 [3] as part of an extensive hydrobiological survey of Sedlice reservoir on the Želivka river. The study was conducted at nine sites at depths of 6–8 m using a trap placed just above the reservoir bottom. After a 24-hour exposure, several dozen water mites were captured at each site. A total of six species were recorded, differing markedly from the species composition of water mites in the water reservoirs presented in this study. Representatives of the genera Unionicola Haldeman, 1842 and Neumania Lebert, 1879 were absent, while the most abundant species were Piona coccinea (Koch, 1836) and Hygrobates longipalpis (Hermann, 1804). The occurrence of Lebertia fimbriata Thor, 1899 is noteworthy, as this species was not found in the studied water reservoirs. Additional species recorded from the genus Piona Koch, 1842 included Piona rotunda (Kramer, 1870), now Piona rotundoides (Thor, 1897), and Piona fallax (Thon, 1899). This species, described by Thon from Munický pond in Czechoslovakia [14], does not appear in current hydrachnological literature and is not listed among the synonyms of existing species of the genus Piona Koch, 1842. Láska [15] included Piona fallax (Thon, 1899) in his list of water mites recorded in the Czech Republic, but it has not appeared in any subsequent studies on the water mite fauna of the country. Viets [16] also mentions this species in his 1955 publication and reports its occurrence in Great Britain. Since the cited record from Sedlice reservoir, however, the species has not been recorded again; its existence therefore remains uncertain and cannot be verified, as the material from Sedlice reservoir is no longer available.
A second record of water mite occurrence from this region comes from Kníničky reservoir in Moravia, where Hrabě [17] studied the colonisation of the littoral zone and reservoir bottom. In samples collected with a plankton net at depths of up to 1.5 m at eight sites, he recorded eight species of water mites. The most abundant species was Hygrobates longipalpis (Hermann, 1804), but he also reported occasional occurrences of Unionicola crassipes (Mueller, 1776), Neumania limosa (Koch, 1836), and Mideopsis orbicularis (Mueller, 1776), which are among the species with the highest frequency of occurrence in the water reservoirs studied here.
A detailed study of water mite fauna was published by Punčochář and Hrbáček [18] from Hubenov reservoir in the Bohemian-Moravian Highlands, where they described the dominance of Piona carnea (Koch, 1836), which occurred in the plankton of the reservoir pelagic zone after the reservoir was filled in 1972. During the first few years, the fish stock consisted solely of brown trout (Salmo trutta fario), while large cladocerans of the genus Daphnia dominated the zooplankton and limited the development of phytoplankton biomass. Water transparency in the reservoir exceeded 9 m (the maximum depth of the reservoir is 16 m). In the water mite fauna in the years 1976–1978, Piona carnea (Koch, 1836) predominated (70–90 %); owing to the dense swimming setae on its legs, it was able to move within the pelagic zone. Complementary species included Piona pusilla (Neuman, 1875) and Piona rotundoides (Thor, 1897), which accounted for 5–17 % of the water mite fauna. A change in fish stock, when trout were replaced due to disease by an ichthyofauna dominated by non-predatory fish, was accompanied by a marked shift in the water mite assemblage: the originally “complementary species” subsequently became dominant, and the occurrence of additional species increased, including Limnesia maculata (Mueller, 1776) and Unionicola crassipes (Mueller, 1776). Monitoring of Hubenov reservoir is also included in the present study (site No. 45 – Tab. 1 and 2, Fig. 2). Its fish stock currently consists of a mixture of species [19], in which predatory fish do not predominate, and the water mite fauna is characterised by a clear dominance of Unionicola crassipes (Mueller, 1776), see Tab. 3. A similar, unusual dominance of the water mite Piona limnetica Biesiadka was described by Gliwicz and Biesiadka [20] in the plankton of a dam reservoir in Panama. As in the case of Hubenov reservoir, the main reason was the availability of planktonic cladocerans as a food source, together with suitable body morphology, as the legs of this species, with a specific arrangement of swimming setae, enable movement (swimming) in open water outside the littoral zone. Species of the genera Neumania Lebert, 1879 and Unionicola Haldeman, 1842 were present in lower numbers in this reservoir; their swimming setae also allow them to occur in the plankton.
Of the 34 water mite species recorded in the studied water-supply reservoirs, 12 species occurred at more than 10 % of sites (Tab. 5). These are species characterised by pronounced locomotory activity in the pelagic zone.
With regard to the occurrence and biology of Unionicola crassipes (Mueller, 1776), reference should be made to the publication by the Czech author Ladislav Halík from 1924 [21], in which he described the “parachute-like movement of individuals of this species”, enabled by extremely long legs bearing numerous swimming setae. This was confirmed by later studies [20, 22]. A similar limb morphology is also characteristic of species of the genus Neumania (Fig. 10). Other abundant water mite species – Brachypoda versicolor (Mueller, 1776), Arrenurus albator (Mueller, 1776), Forelia liliacea (Mueller, 1776), and Hygrobates longipalpis (Hermann, 1804) – also possess dense swimming setae on their legs. They commonly occur in the littoral zone with vegetation in standing waters, although they do not reach such a high relative abundance there [40].
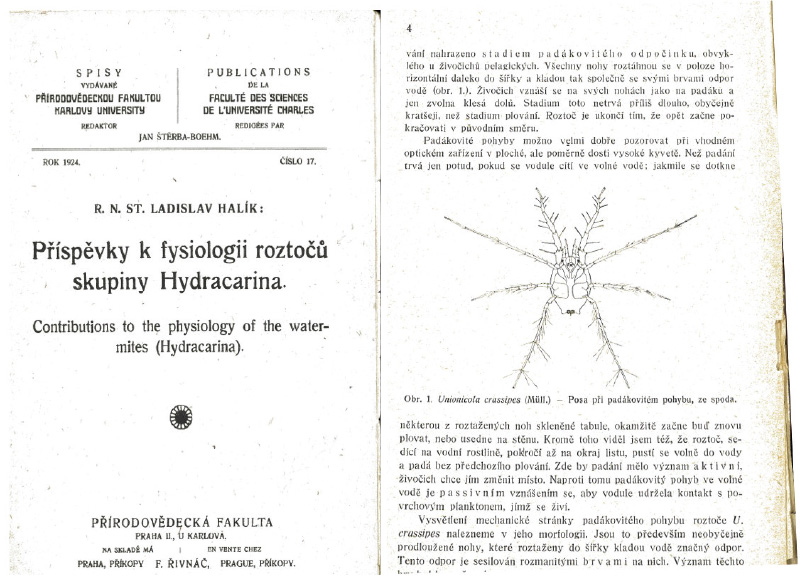
Fig. 10. Example of a publication by Halík from 1924 [21] (title page and drawing of Unionicola crassipes (Mueller, 1776)); the text describes the locomotor activity of this water mite species in the water column: swimming alternately with parachute-like descent with the long legs with swimming setae extended
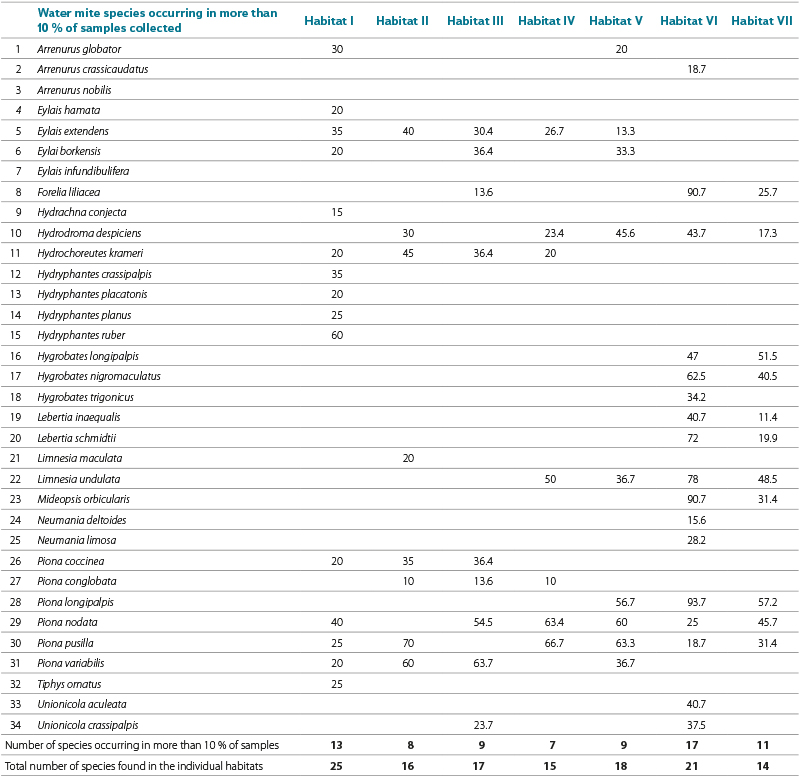
Unfortunately, in most studies of reservoir ecosystems, information on water mite fauna is only general [23] or their presence is referred to merely as a group of organisms [24]. A very similar composition of dominant water mite species in littoral habitats with and without macrophyte vegetation was reported from Rybinsk reservoir by Tuzovskij [13], as shown in Tab. 7. In habitats with macrophyte vegetation, he found a markedly different species composition of water mites, with a predominance of species of the genus Piona Koch, 1842 and others, which are also common in ponds [33, 40].
Tab. 7. The occurence of water mite species found in more than 10 % of samples in seven littoral biotops of the Rybinsk reservoir. Data taken from the publication Tuzovskij [13], who investigated the fauna of water mites in different littoral biotops of the Rybinsk reservoir in samples taken by a grab-sampler in 1970–1971 (total number of samples 802). A brief description of biotopes: Biotope I – The most flooded zone of the littoral with the predominance of Carex acuta L., Carex vesicaria L., Carex nigra (L.) Reich. Biotope II – Rorripa amhibia (L.) stands on areas of 30–40 m2 they closely follow the previous Carex stands. Biotope III – Reed community (Phragmites communis Trin.) on a sandy bottom, with an insignificant occurrence of other plants. Biotope IV – Potamogeton heterophyllus Schreib. stands with a relatively significant admixture of Agrostis stolonifera L. and Eleocharis acicularis (L.) Roem. et Schult. Biotope V – Eleocharis palustris (L.) stands with admixture of Alisma plantago-aquatica L., Potamogeton heterophyllus Schreib., Eleocharis acicularis (L.) Roem. et Schult., and others. Biotope VI: Protected coast, gray clay bottom, no vegetation, depth up to 2.5 m. Biotope VII: Surf zone of the coast of Khokhotki Island, sandy bottom, no vegetation
A, B – Hygrobates trigonicus (Koenike, 1895); [A] male; [B] female (preparation nr. 41-2).
C, D, E – Brachypoda versicolor (Mueller, 1776); [C] male; [D] male, side view; [E] female; (preparations nr. 2-1 and nr. 15-3).
F, G – Arrenurus albator (Mueller, 1776); [F] male, [G] female (preparation nr. 29-2).
H – Forelia longipalpis (Maglio, 1924); female (preparation nr. 41-1).
I – Unionicola crassipes (Mueller, 1776); female (preparation nr. 45-2).
J – Neumania limosa (Koch, 1836); male (preparation nr. 35-2).
K – Unionicola crassipes (Mueller, 1776); female (preparation nr. 7-2).
L – Neumania limosa (Koch, 1836); male (preparation nr. 16-4).
M, N – Forelia liliacea (Mueller, 1776); [M] male (preparation nr. 3-4), [N] female (preparation nr. 8-8).
O – Vicinaxonopsis (Cook, 1974); female, not determined to species level (preparation nr. 37-4).
Microphotographs of several water mite species found in the investigated reservoirs. The first digit in the specimen designation is the locality number according to Tab. 1 and 2, the second digit is number of preparation prepared from this locality. All bars are 0.5 mm.
There is a substantial body of detailed publications on the fauna of water mites in lakes. A comparison of the results from drinking water reservoirs presented in this study with data from lakes is difficult and problematic, as the sampling methods used differ considerably. For monitoring the occurrence and composition of water mite fauna in lakes, various quantitative sampling methods have been employed, including traps [30], dredges [12, 13], and grabs for benthic sampling [25, 27]. At the same time, sampling sites were predominantly located in the littoral zone with macrophyte vegetation, i.e. in a habitat similar to that found in fishponds. In a quantitative study of water mites in 50 lakes in Schleswig-Holstein, Viets [27] reported 58 species of water mites in samples collected using a grab. Thirteen species occurring at more than 20 % of sites include seven species with the highest frequency of occurrence in the drinking water reservoirs studied – Unionicola crassipes (Mueller, 1776), Hygrobates longipalpis (Hermann, 1804), Forelia liliacea (Mueller, 1776), Neumania deltoides (Piersig, 1894), Mideopsis orbicularis (Mueller, 1776), Arrenurus albator (Mueller, 1776), and Arrenurus crassicaudatus Kramer, 1875 (Tab. 5). These water mite species also occur most frequently in Dutch lakes [26].
In the oligotrophic lakes of Stechlinsee [12] and Bodensee [25], in samples taken from the bottom using a benthic grab and dredge, the species Brachypoda versicolor (Mueller, 1776), Unionicola crassipes (Mueller, 1776), and Hygrobates longipalpis (Hermann, 1804), which dominate in drinking water reservoirs, were among the most frequent. By contrast, Arrenurus albator (Mueller, 1776) occurred only sporadically in Stechlinsee and was completely absent from Bodensee. In both lakes, however, species of the genus Lebertia Neumann, 1880 were present.
Biesiadka [28] compared the composition of water mite fauna from the littoral zone of Lake Kierskie in collections from the period 1930–1933 with results from 1969–1970. Although the composition of the water mite fauna changed slightly, the dominant species were the same as those in drinking water reservoirs, with the exception of a notable occurrence of Lebertia insignis Neumann, 1880.
The composition of water mite fauna from 21 oligotrophic reservoirs (with chlorophyll a concentrations ranging from 0.22 to 4.81 µg/L) was published by Pozojevič et al. [29]. The genus Lebertia Neumann, 1880 (frequency of occurrence 57 %) and the species Arrenurus albator (42 %), Neumania deltoides (47 %), and Hydrochoreutes krameri Piersig, 1896 (38 %) formed a common component of the water mite fauna. The authors do not report the occurrence of Hygrobates longipalpis (it may be included under the genus Hygrobates Koch, 1837, which they did not specify in more detail). The species Brachypoda versicolor (Mueller, 1776) was recorded at only two sites (9 %). A more detailed comparison of species composition with the results from the studied reservoirs is not possible, as the occurrence of the genera Limnesia Koch, 1836, Neumania Lebert, 1879, and Hydrodroma Koch, 1837 is likewise reported without species-level identification. By contrast, Unionicola crassipes (Mueller, 1776) was not recorded, which is surprising, as it commonly occurred in lakes (particularly oligotrophic ones) [12, 13, 22, 25–28, 30, 37]. Viets [27] states that an essential condition for the occurrence of this water mite species is the presence of bryozoans. Unfortunately, Pozojevič et al. [29] and most of the other cited references do not provide more detailed information on the fauna of the studied lakes. The occurrence of this species is probably strongly influenced by habitat conditions that allow individuals to move/swim (suspend) in the open water, which is, of course, limited by dense stands of macrophyte vegetation. In all the oligotrophic lakes mentioned, species of the genus Lebertia Neumann, 1880 were present, which Lundblad [10, 11] reported as a common component of the water mite fauna of Swedish lakes. Representatives of this genus were not found in any of the studied drinking water reservoirs, which represents the main fundamental difference from the species composition of water mite fauna in lakes.
The cited results from lakes [10, 11, 27] were obtained from benthic samples collected using a grab or dredge at depths greater than 2–4 m (in some cases even 10 m or more), where species of the genus Lebertia Neumann, 1880 commonly occurred. This is also confirmed by data from Sedlice reservoir [3], in which water mites of this genus were found just above the bottom (in a trap placed 5 cm above the sediment surface), in the sublittoral zone at depths of 6–8 m. Pieczynski [30], in his extensive study of water mites in the littoral zone of Polish lakes, sampled using traps placed in vegetated littoral habitats, and reported virtually the same species that dominated in drinking water reservoirs, but accompanied by additional water mite species typical of the littoral of standing waters. However, he only recorded representatives of the genus Lebertia Neumann, 1880 in a single lake, in a habitat influenced by the inflow of a watercourse. In the littoral zones of the other studied lakes, he did not report the occurrence of species of the genus Lebertia.
A probable reason for the absence of species of the genus Lebertia Neumann, 1880 in the studied drinking water reservoirs is the character of the habitats from which the samples were taken. The author of the study recorded water mites of the genus Lebertia Neumann in hand-net samples from the shallow littoral of the oligotrophic Lake Ohrid at depths of up to 1 m (unpublished data). Thus, the sampling method itself does not necessarily have a decisive influence on the occurrence of water mites of this genus, although species of this genus clearly show a preference for habitats at greater depths.
It is evident that not only the character of the habitat, but also the methods used for collecting water mites, have a significant influence on both the occurrence and abundance of individual species. In this study, the occurrence of individual water mite species is compared with published data without assessing the effect of the methods used, which differ considerably – from passive “traps” to quantitative sampling per unit area of the bottom (using corers, grabs, or dredges). The comparisons presented therefore rely on data on the most frequent occurrence of individual species in the littoral zones of different sites.
The abundance of water mites in individual drinking water reservoirs shows considerable variation (Tab. 3), with the highest numbers recorded in Nová Říše reservoir (site No. 40; see Fig. 9) and Vrchlice reservoir (site No. 3). This may be due to the short distance of the sampling sites from macrophyte stands in the littoral zones of both reservoirs, as aquatic vegetation provides a range of favourable conditions for water mite communities, as evidenced by data in [22, 30, 32]. In Josefův Důl reservoir (site No. 4), no water mites were recorded, while in Souš reservoir (site No. 5) only four nymphs were found (one of which could be identified as a representative of the genus Hydrochoreutes Koch, 1837). The reason is evidently the low pH of water in these reservoirs, which fluctuates considerably over the course of the year: outside the summer season, values drop to around 5.0–5.5, whereas in summer they reach 6.0–7.0, as documented by long-term water quality monitoring carried out by the Elbe River Board, state enterprise. The effect of low pH values on the absence of water mites was also described, for example, by Lundblad [10] in Swedish lakes.
The methodology used for studying water mite fauna does not provide absolute quantitative data; however, it allows comparisons of fauna from the same habitat across individual sites. The range of chlorophyll a concentrations recorded in the studied drinking water reservoirs is relatively narrow (indicating oligotrophic to slightly mesotrophic conditions), which limits the possibility of drawing conclusions about relationships with water mite diversity. Nevertheless, a comparison of water mite occurrence among reservoirs with different chlorophyll a concentrations suggests a tendency towards higher abundance and greater species richness in reservoirs with concentrations below 20 µg/L (Nos. 34 – Karolinka, 14 – Žlutice, 8 – Husinec) compared to reservoirs with concentrations above 20 µg/L (Nos. 2 – Křižanovice, 20 – Podhora, 36 – Fryšták).
Current literature emphasises the potential of water mites as indicators of the state of aquatic ecosystems [34], as, through their dependence on larval hosts and trophic interactions, they integrate the character of the biotic community. The relationship between water mite community diversity and water quality has been studied primarily in running waters [35, 36]. The occurrence of water mites in relation to reservoir trophic status has not yet been demonstrated [30, 37], since most available data on water mites from lakes originate from the littoral zone, which is a highly heterogeneous habitat where the abundance of organisms and the representation of different communities within the aquatic ecosystem are influenced by a range of additional factors.
The importance of littoral habitats in lakes for the occurrence of different water mite species is demonstrated by the cited study of Tuzovský [13] and the data presented therein, summarised in Tab. 7. Verification and evaluation of the influence of different littoral habitats in specific reservoirs would undoubtedly enhance our understanding of the indicative potential of water mite diversity in standing waters (both reservoirs and lakes).
The occurrence of water mites is influenced by a range of abiotic and biotic factors, with the presence of hosts for their developmental stages (larvae) being particularly important, as well as the availability of food [22, 38], the presence of predators, and the hydrochemical and hydromorphological characteristics of the water body. For this reason, the species composition of water mite fauna can be considered one of the key integrative indicators of the state of an aquatic ecosystem, although it may not always be clear which of these factors is dominant or decisive. The presented results and their comparison with data from the literature indicate that habitat plays a significant role, as it can enhance the representation of species better adapted to particular conditions, even though these species may be absent from sites with a different habitat within the same water body. Together with the relatively difficult identification of water mite species, this represents one of the main reasons for the still limited use of water mite fauna in the assessment of aquatic ecosystems.
CONCLUSION
In samples of water mite fauna collected from the stony littoral of 45 drinking water reservoirs in the Czech Republic in August and September 2024, a total of 1,356 water mites were recorded (849 adults and 507 nymphs). A total of 34 water mite species were identified, while one specimen (probably belonging to the genus Vicinaxonopsis Cook, 1974) has not yet been determined to species level. Twelve water mite species were recorded at more than 10 % of sites: Brachypoda versicolor (Mueller, 1776), Unionicola crassipes (Mueller, 1776), Arrenurus albator (Mueller, 1776), Forelia liliacea (Mueller, 1776), Neumania limosa (Koch, 1836), Neumania deltoides (Piersig, 1894), Hygrobates longipalpis (Hermann, 1804), Arrenurus sinuator (Mueller, 1776), Mideopsis orbicularis (Mueller, 1776), Arrenurus crassicaudatus Kramer, 1875, Hydrodroma despiciens (Mueller, 1776), and Neumania vernalis (Mueller, 1776). These species are characterised by high locomotory activity (swimming in open water), enabled by swimming setae on their legs. The representation of these species accounted for 87.4 % of all recorded individuals. Species of the genus Neumania Lebert, 1879 and especially Unionicola Haldeman, 1842 have very long legs and can therefore be considered typical inhabitants of the so-called “lacustrine zone” of reservoirs, i.e. the area in front of the dam, although their ontogenetic development and trophic relationships are linked to the morphology of the shoreline and bottom. These species also occur most frequently in lakes, where, however, representatives of the genus Lebertia Neumann, 1880 are also among the most common species, although they were not found in the studied drinking water reservoirs. The reason is probably the sampling methodology using a plankton net, which captures the occurrence of water mites above the stony bottom to a depth of 1.0 m, whereas data from lakes were obtained from benthic samples collected using a grab or dredge at depths greater than 1 m. Three recorded species – Arrenurus albator (Mueller, 1776), Hygrobates trigonicus Koenike, 1895, and Forelia longipalpis Maglio, 1924 – have extended the checklist of water mites of the Czech Republic maintained by the Nature Conservation Agency of the Czech Republic. Forelia longipalpis Maglio, 1924 represents a species new to the fauna of the Czech Republic, as its occurrence has not previously been reported in the literature.
Acknowledgements
The author would like to express sincere thanks to the Directors General of all River Boards, state enterprises for granting permission to carry out sampling in the drinking water reservoirs under their management – namely Petr Kubala from the Vltava River Board, state enterprise, Marián Šebesta from the Elbe River Board, state enterprise, Jan Svejkovský from the Ohre River Board, state enterprise, Václav Gargulák and the current Director General David Fína from the Morava River Board, state enterprise, as well as Jiří Tkáč and the current Director General Petr Birklen from Odra River Board, state enterprise.
At the same time, I would like to thank all workers operating dams of drinking water reservoirs in the Czech Republic, whom I had the pleasure of meeting in person during the sampling campaigns, and who enabled access to the dam structures and, in many cases, kindly assisted with sampling when access to the water surface from the dam was difficult.
I am greatly indebted to the staff of the water management laboratories of all state-owned River Boards, state enterprises, for providing data from the monitoring of drinking water reservoir quality, which made it possible both to compare the studied sites and to interpret the occurrence of certain water mite species in comparison with European lakes.
I also thank my colleague Monika Stádníková for preparing a clear map of the distribution of existing drinking water reservoirs within the Czech Republic.
For assistance in obtaining sources of foreign literature, thanks are due to Prof. Jan Kubečka from the Institute of Hydrobiology of the Biology Centre of the Czech Academy of Sciences, and also to Ing. Piotr Barański, Head of the Secretariat of the International Commission for the Protection of the Oder against Pollution (Wroclaw, Poland).
I also thank the management of the Nature Conservation Agency of the Czech Republic for supporting this work through a grant for the expansion of the “Species Occurrence Database of Nature Conservation”, which will be enriched with data on the occurrence of water mites in reservoirs.
I am very grateful to Ing. Josef Nistler and Mgr. Zuzana Řehořová (both from TGM WRI) for enabling the publication of this article in the VTEI Journal and for their assistance with the final formal editing of the manuscript.
The Czech version of this article was peer-reviewed, the English version was translated from the Czech original by Environmental Translation Ltd.